
The Pilot COVID-19 At-Home Test, distributed by Roche Diagnostics, is previously known as the COVID-19 At-Home Test. Video: What Is an EUA? Describes how the FDA can issue an emergency use authorization (EUA) to provide more timely access to diagnostic tests that may help during the public health emergency when there is no adequate, approved, and available alternative.Isolation and Precautions for People with COVID-19 | CDC.What to Do If You Were Exposed to COVID-19 | CDC.COVID-19 Self-Testing At-Home or Anywhere | CDC.COVID-19 Testing: What You Need to Know | CDC.Video: How to Interpret Self-Test Results | CDC.Counterfeit At-Home OTC COVID-19 Diagnostic Tests.At-Home COVID-19 Antigen Tests-Take Steps to Reduce Your Risk of False Negative: FDA Safety Communication. 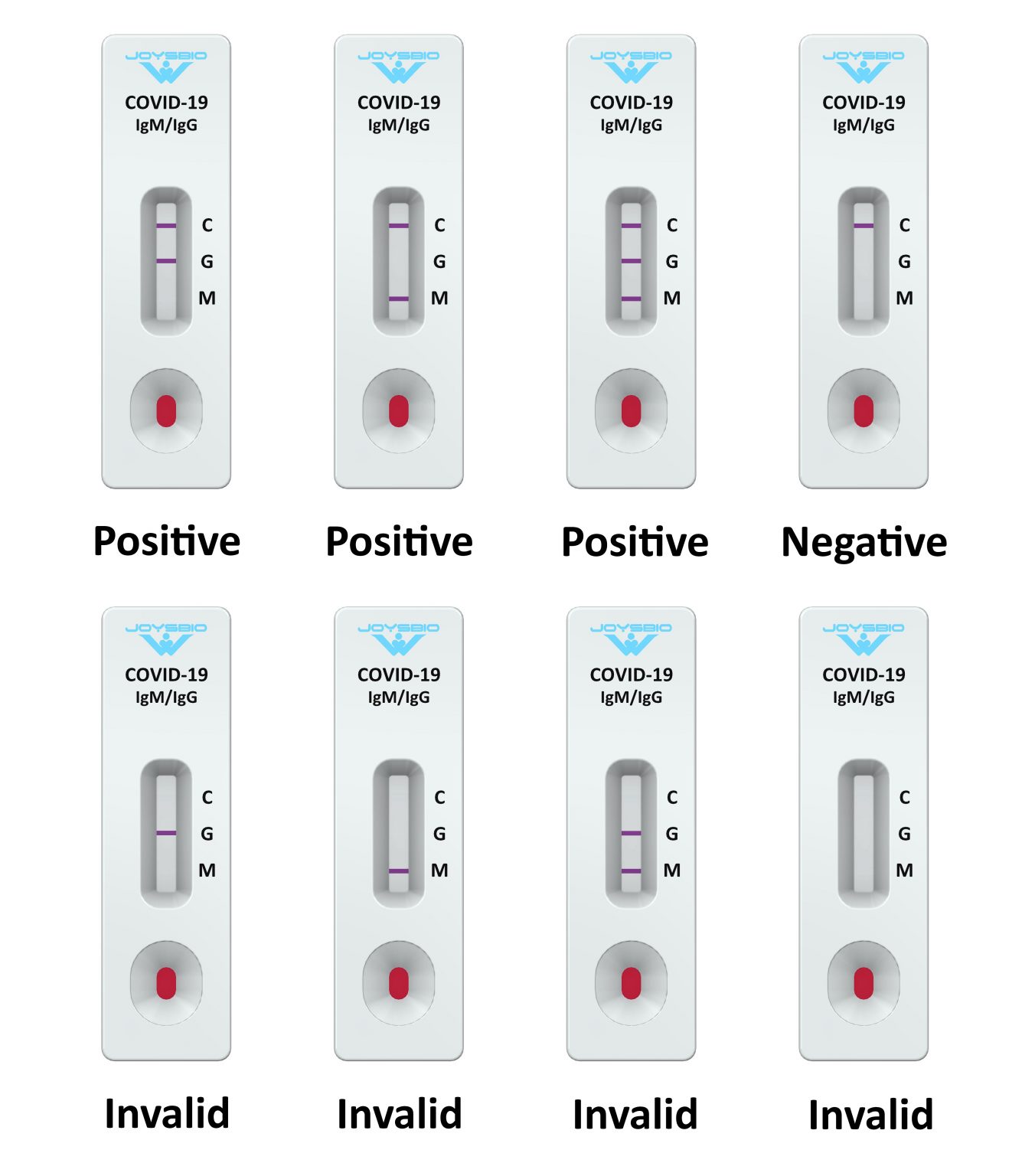
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
